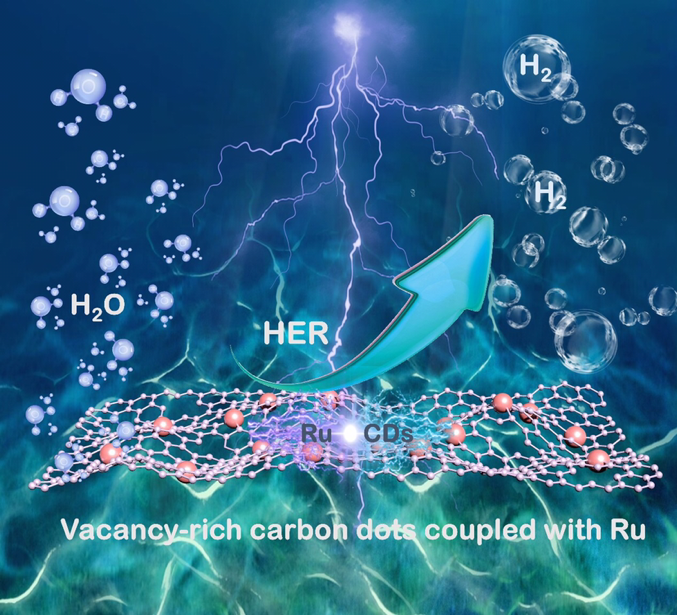
HIT News—Recently, Professor Li Baoqiang’s research group from Academician Zhou Yu’s team at the School of Materials, by associating Ruthenium(Ru) with vacancy-rich Carbon Dots(CDs), construct Ru and vacancy-rich Carbon Dots electrocatalysts(Ru@CDs). The research also demonstrates the mechanism of strong electron coupling for enhanced electro-catalyzed hydrogen evolution activity. Their findings not only expand the applications of CDs, but also provide fundamental insights for new-type hydrogen evolution electrocatalysts with high efficiency. This paper was published in “Small”, with the title of “Strong Electron Coupling of Ru and Vacancy-rich Carbon Dots for Synergistically Enhanced Hydrogen Evolution Reaction”. Lin Zonglin, 2019 PhD student in the School of Materials, is the first author of the paper. Harbin Institute of Technology is the first corresponding unit. And Professor Li Baoqiang is the first corresponding author.
New energy is the hot topics of research in the field of industry and academia, among which hydrogen energy is currently recognized as clean energy. The electrolysis of hydrogen depends on high-performance electrocatalysts. Ruthenium(Ru), with its low cost (about 5% of Pt), is considered as an ideal alternative to platinum(Pt)-based electrocatalysts owing to their similar hydrogen bond strengths. However, its activity is still not satisfactory. What’s more, the tendency of Ru to aggregate due to its high cohesive energy degrades its activity. Vacancy engineering is an excellent strategy for modulating the physicochemical and electronic properties of nanomaterials to improve their electrocatalytic activity. However, creating vacancies directly into pure metallic Ru is difficult. Therefore, appropriate means to overcome this problem are required to provide rich vacancies. CDs, which possess abundant intrinsic vacancies, rapid electron-transfer properties, high stability, and large surface area, are ideal candidates for combination with Ru to design a new-type electrocatalyst. Additionally, CDs, with abundant vacancies and functional groups, can provide favorable binding sites for the coordination stabilization of metal-CDs composites by anchoring and trapping. And the confinement of CDs prevents the aggregation of Ru nanoparticles and improves its stability.
To overcome low activity and aggregation of Ru catalyst, Professor Li Baoqiang’s research team propose and realize the combination of Ru with vacancy-rich carbon dots, as Ru@CDs, which elaborately introduce vacancies in a Ru composite structure, efficiently reduce the instability and aggregation of Ru nanoparticles, thus enhancing its electrocatalytic activities. As expected, Ru@CDs exhibited excellent catalytic performance with a low overpotential of 30 mV at 10 mA cm-2 in 1 M KOH, a small Tafel slope of 22 mV decade-1. Its electrocatalytic activities is comparable to those of most previously reported Ru-based electrocatalysts and commercial Pt/C catalyst. Additionally, spectroscopic characterizations and theoretical calculations demonstrate that the rich vacancies and the electron interactions between Ru and CDs synergistically lower the intermediate energy barrier and thereby maximize the activity of the Ru@CDs electrocatalyst.
Link to full article: https://doi.org/10.1002/smll.202102496
Editor: Zhang Yan