
Recently, the Institute of Modern Microscopic Instruments in the School of Instrumentation Science and Engineering of HIT has made breakthrough in the field of optical super-resolution microscopic imaging technology. Under the condition of low phototoxicity, the research team improved the resolution of structured light microscope from 110 nm to 60 nm, realizing long-term, ultrafast and live-cell super-resolution imaging. On November 16, the research results, with the title of “Sparse Deconvolution Improves the Resolution of Live-cell Super-resolution Fluorescence Microscopy”, were published online in the form of a long article in Nature Biotechnology, an international authoritative journal (Nature Biotechnology, with an impact factor of 54.908 in 2020, only publishes engineering technologies with major breakthroughs in the field of life sciences).
The research results are mainly completed through the cooperation between the School of Instrumentation Science and Engineering of HIT and the School of Future Technology of Peking University. HIT is the first unit of the thesis. Zhao Weisong, doctoral student in HIT, and Zhao Shiqun and Li Liuju, post doctorates in Peking University, are the co-first authors of the thesis. Associate Professor Li Haoyu of HIT and Professor Chen Liangyi of Peking University are the co-corresponding authors of the thesis. Professor Liu Jian and Academician Tan Jiubin of HIT are the co-authors of the thesis and the heads of the scientific research team of HIT. Cooperation units also include the National Nano Center of the Chinese Academy of Sciences, the Institute of Biophysics of the Chinese Academy of Sciences, and Wuhan University, etc.
The resolution of microscopic instruments represents the boundary of human’s scientific exploration. The 2014 Nobel Prize in chemistry was awarded to three scholars who had made remarkable achievements in super-resolution fluorescence microscopy. The research team of Modern Microscopy Instruments of HIT proposed a computational microscopic imaging algorithm that can break through the optical diffraction limit, using the forward physical model of fluorescence imaging and the theory of compressed sensing, combining the dual constraints of sparsity and space-time continuity, and establishing a general solution framework called sparse deconvolution technology, which breaks through the hardware limitation of the existing optical super-resolution microscopic system and expands the spatial-temporal resolution and spectrum.
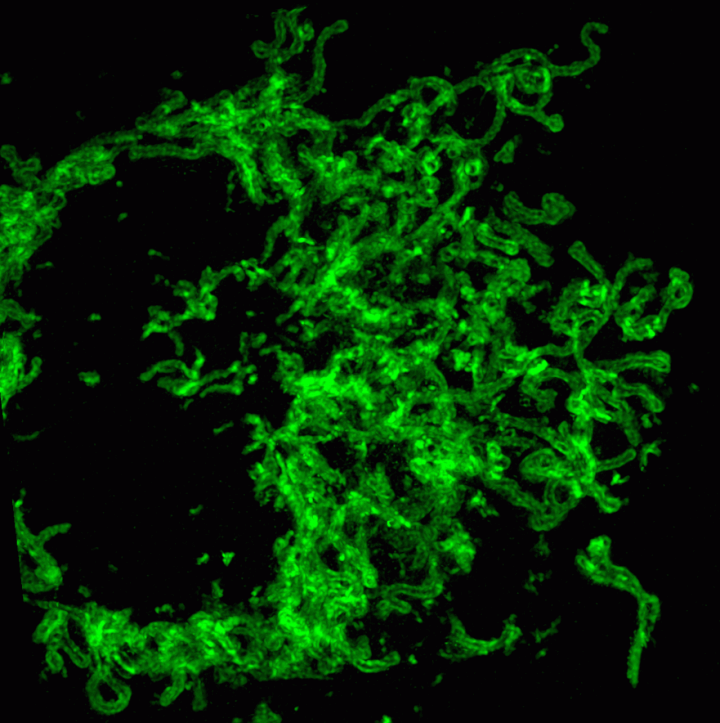
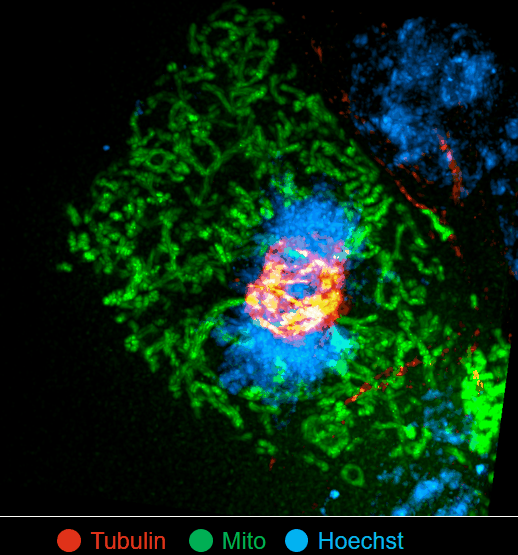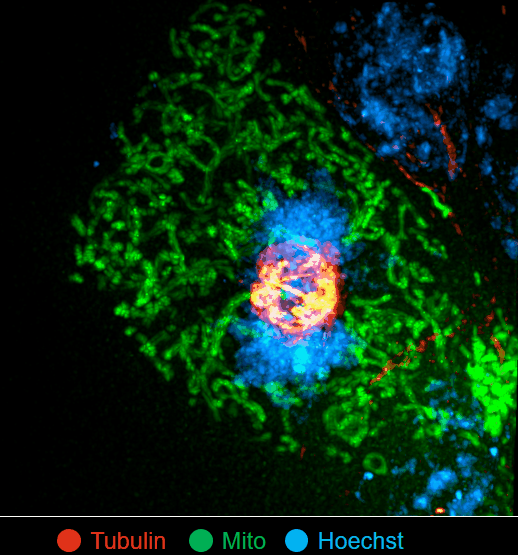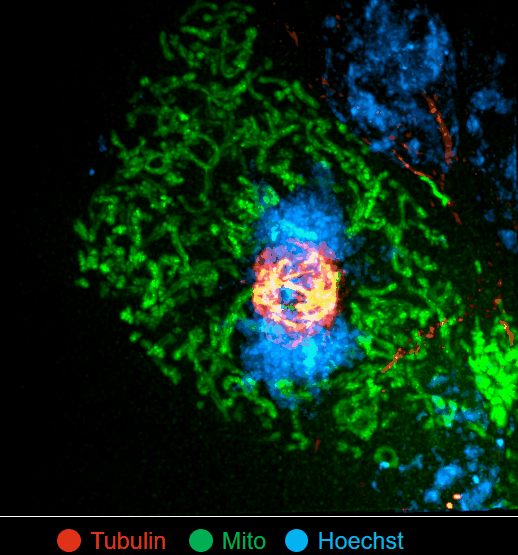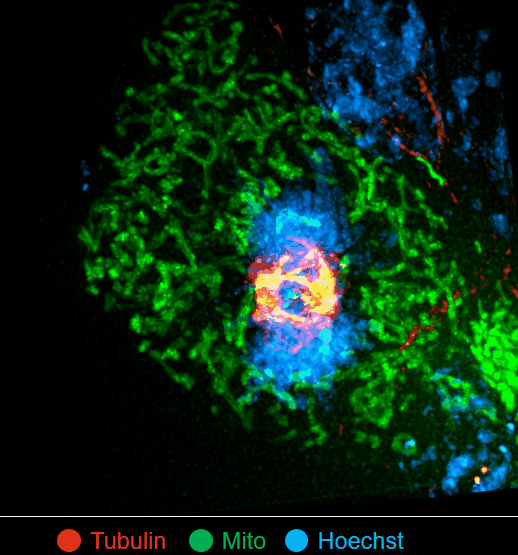
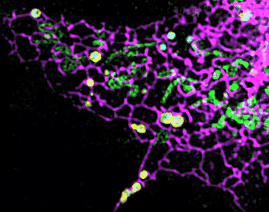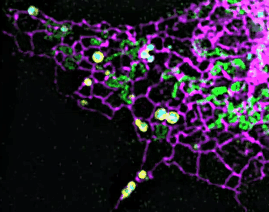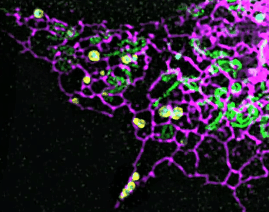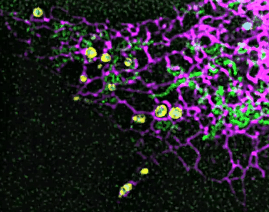
On this basis, the research team has developed the ultrafast structured light super-resolution fluorescence microscope system (Sparse-SIM). The system is featured by super-resolution, high-throughput, non-invasion and low-toxicity. Under the condition of high-speed imaging, it has a resolution better than 60 nm and ultra long-time live cell dynamic imaging performance of more than 1 hour. The team is the first to observe the fusion pore with two characteristics in the process of pancreatic islet secretion, and for the first time, they observed by using the linear structured light microscope the circular nuclear pore complex and caveolin labeled with different proteins that can be distinguished only under nonlinear conditions. In addition, the researchers also demonstratedthe process of analyzing the actin dynamic network, and the rapid behavior of lysosomes and lipid droplets deep in cells by using the imaging technology, and recorded the fine relative movement between the inner and outer membranes of double-color mitochondria.
Based on physical and chemical methods, this research proposed for the first time a general model that breaks the optical diffraction limit from the perspective of calculation, and realized the great principle innovation. It is asuper-resolution microscopic instrument with the highest resolution (60 nm), the fastest speed (564 frames/sec) and the longest imaging time (more than 1 hour) in the current live-cell optical microscopic imaging. The technical framework has also proved to be applicable to most current fluorescence microscopic imaging system modes, and can achieve nearly twice the stable spatial resolution improvement, providing a new generation of biomedical super-resolution imaging instruments for precision medicine and novel drug research and development, and thus making it possible to greatly accelerate the high-precision characterization of disease models in the future.
Link to full article:https://www.nature.com/articles/s41587-021-01092-2


