A research group, under the guidance of Academician Ma Jun and led by Professor Wang Wei from the School of Environment at Harbin Institute of Technology (HIT), has made significant progress in low-energy, chemical-free green water treatment. Their study, entitled Ultrafast energy-neutral molecular oxygen activation via atomically adjacent bimetallic catalytic sites, has been published in Nature Communications.
As the world's most abundant natural oxidant, dioxygen (O2) is regarded as an ideal alternative to conventional chemical oxidants such as hydrogen peroxide, persulfate, and ozone. However, the triplet ground state of O2 imposes intrinsic spin-forbidden constraints, leading to sluggish electron transfer kinetics and significant reaction barriers.
To overcome these limitations, existing O2 activation strategies typically rely on external energy inputs such as light, electricity, or heat, which compromise economic feasibility and sustainability. In contrast, energy-neutral O2 activation systems often suffer from isolated active sites, localized electronic states, and cumulative multi-step electron-transfer barriers, resulting in low rates of reactive oxygen species (ROS) generation, insufficient catalytic activity, and rapid intermediate dissipation, limiting practical applicability.
Addressing this scientific and technological challenge, Wang’s group proposed a novel strategy for molecular oxygen activation that requires neither external oxidants nor additional energy input. By constructing atomically adjacent bimetallic catalytic sites, the team leveraged d-d orbital coupling and electron delocalization to establish short-range, highly efficient electron-transfer pathways within the catalyst. This design enhances O2 adsorption and facilitates effective weakening of the O-O bond through π* orbital interactions, enabling direct and selective conversion of O2 into singlet oxygen (1O2) under mild conditions.
Under bias-free conditions with simple aeration, the system achieved ROS generation rates and pollutant degradation performance comparable to those of conventional chemical oxidation processes, while reducing operational costs by approximately two orders of magnitude. Moreover, the catalyst demonstrated excellent stability and versatility in complex aqueous matrices, exhibiting broad applicability for organic pollutant degradation, inorganic ion transformation, and bacterial inactivation. This work establishes a new design paradigm for sustainable and efficient molecular oxygen activation and lays a solid foundation for the development of next-generation energy-neutral catalytic technologies.
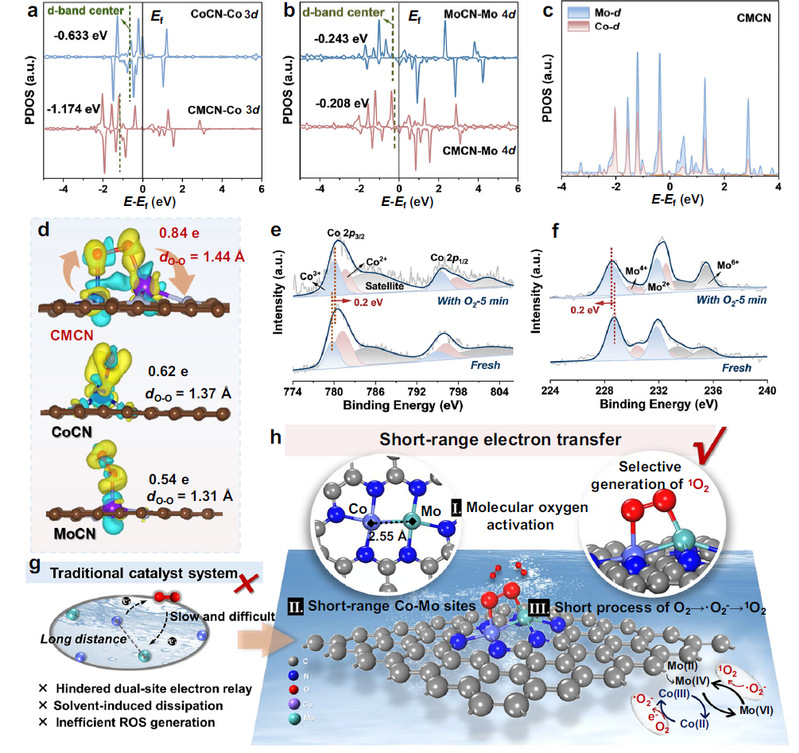
The activation mechanism of molecular oxygen electron transfer driven by adjacent dual-site cooperation. [Photo/hit.edu.cn]
HIT is the first corresponding institution of the paper, with Chen Xi, a doctoral candidate from the School of Environment, as the first author. Associate Researcher Wang Aiwen, Professor Liu Dongmei, and doctoral student Cao Yang are co-authors. Professor Wang serves as the corresponding author, Professor Liu Xianwei from the University of Science and Technology of China is the co-corresponding author, and Academician Ma provided important guidance throughout the study.