Professor Zhang He, a member of Professor Zhao Jie's research team at the School of Mechatronics Engineering, Harbin Institute of Technology (HIT), has achieved a significant breakthrough in minimally invasive otologic surgical robotics.
The team has developed an inner ear surgical robot system utilizing the ear canal as the surgical access route. The system overcomes critical technical challenges associated with dexterous manipulation, intelligent perception, and precise control of surgical robots within the narrow and complex anatomical structures of the human body. It enables minimally invasive inner ear drug injection and perilymph sampling procedures. When integrated into gene therapy, the system has the potential to enable non-invasive postoperative treatment of congenital hearing loss.
The findings have been published in the internationally renowned journal Nature Communications under the title Interaction-aware Dexterous Robot for Minimally Invasive Transcanal Inner Ear Interventions. This research establishes a novel technological pathway and platform for both fundamental research and clinical translation in the field of inner ear diseases. It is anticipated to drive a transformative advancement toward non-invasive postoperative outcomes in inner ear surgery, and to significantly enhance diagnostic and therapeutic capabilities for inner ear functional disorders, congenital hearing loss, and related conditions.
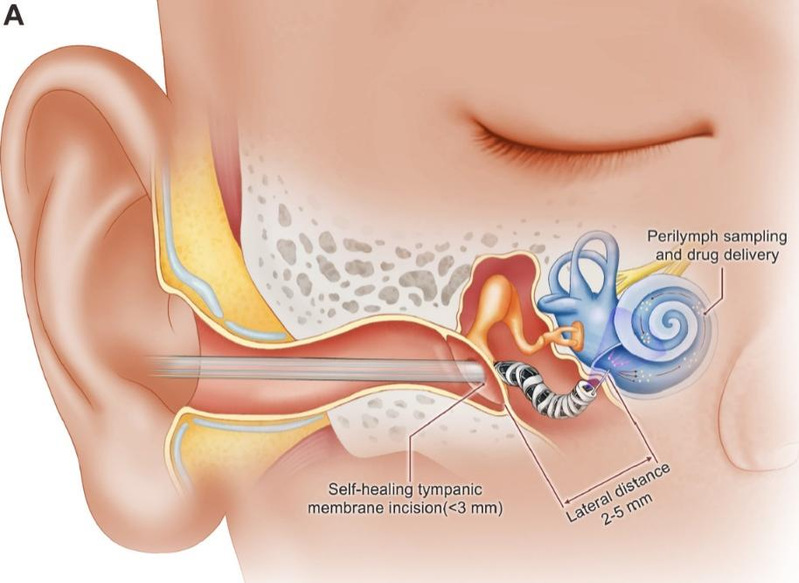
The DS-MDCR (Dual-Segment Miniature Dexterous Continuum Robot) accesses the inner ear through a self-healing tympanic membrane incision, navigates through narrow anatomical structures via spatial S-shaped curvilinear pathways, and precisely reaches the round window membrane to perform targeted puncture, microsampling, and drug delivery. [Photo/hit.edu.cn]
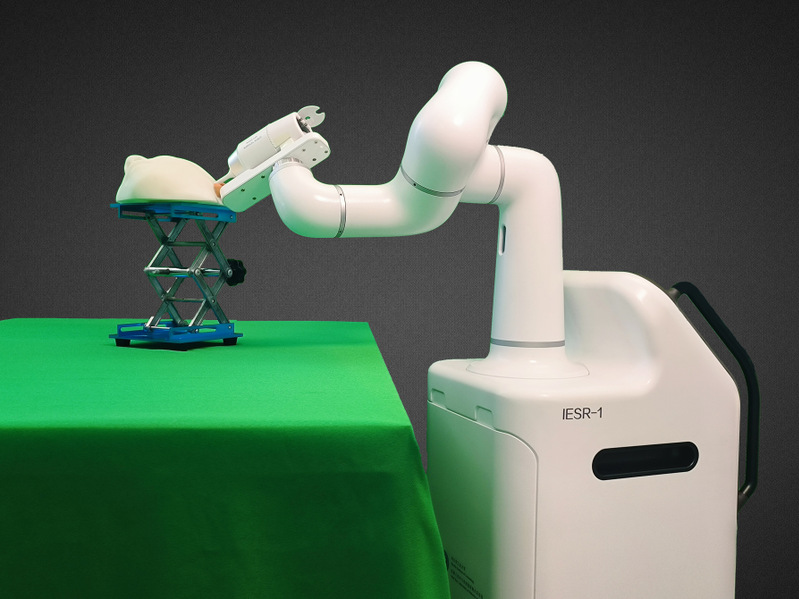 The overall system configuration of the DS-MDCR. [Photo/hit.edu.cn]
The overall system configuration of the DS-MDCR. [Photo/hit.edu.cn]
Inner ear disorders – including sudden sensorineural hearing loss, Meniere's disease, and tinnitus – significantly impair patients' quality of life. As the inner ear is deeply embedded within the temporal bone, diagnosis and treatment have long been hindered by three persistent challenges: difficulty of access, difficulty of sampling, and difficulty of precise intervention. Among existing treatment modalities, systemic drug administration is limited by the blood-labyrinth barrier, while local drug delivery suffers from insufficient tissue penetration and rapid drug dissipation. Compounding these challenges, the ear canal and middle ear present a confined space with tortuous pathways and considerable inter-individual anatomical variation, making it difficult for conventional instruments to perform high-precision procedures while ensuring patient safety. There is therefore a critical need for next-generation minimally invasive surgical devices that combine miniaturization, dexterous manipulation, and tissue interaction sensing capabilities.
To address these challenges, this study presents a low-aspect-ratio dual-segment continuum robot that integrates guidance, visualization, and manipulation into a unified platform. The robot features a saddle-joint structure with no transitional joints, achieving – within a compact form factor of 2 mm outer diameter and less than 7 mm in length – a maximum planar bending angle of ±136° and a minimum bending radius of 1.9 mm. It supports programmable C-shaped and S-shaped three-dimensional configurations, enabling superior adaptability to the complex anatomical pathways of the inner ear. The system is further equipped with a microneedle module integrated with fiber Bragg grating sensors, establishing an independent puncture degree of freedom and real-time force feedback capability. This enables microneedle positioning accuracy of 17.9 ± 4.1 μm, effective sensing of tool-tissue interaction states, and enhanced procedural safety and success rates.
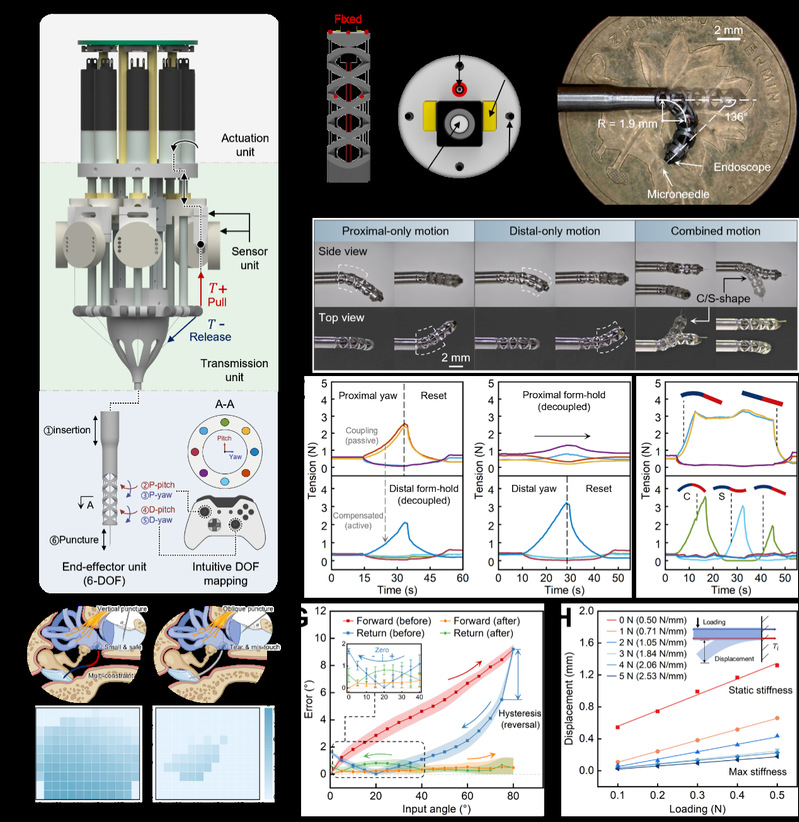 The design and performance characterization of the DS-MDCR. [Photo/hit.edu.cn]
The design and performance characterization of the DS-MDCR. [Photo/hit.edu.cn]
Results demonstrated that the robot performed targeted round window membrane puncture, inner ear fluid sampling, and intracochlear injection via the external auditory canal approach, successfully avoiding the invasive surgical access routes associated with conventional procedures. Animal studies revealed no significant postoperative complications, with no substantial changes in hearing thresholds. Both the round window membrane puncture site and the tympanic membrane incision exhibited progressive healing, confirming the efficacy and safety of this technical approach. The researchers noted that the system holds further potential for integration with teleoperation, augmented reality navigation, and intelligent perception technologies, with the aim of broadening its applicability in primary healthcare settings and precision medicine scenarios.
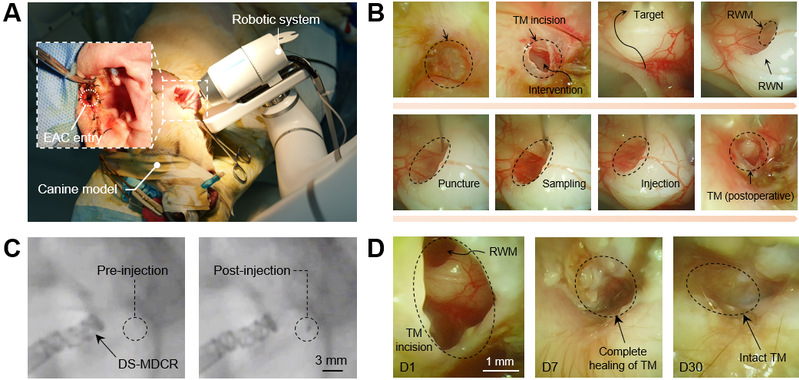 The large animal (canine) experimental validation and tympanic membrane healing assessment. [Photo/hit.edu.cn]
The large animal (canine) experimental validation and tympanic membrane healing assessment. [Photo/hit.edu.cn]
The video summary of overall research contributions. [Photo/hit.edu.cn]
The State Key Laboratory of Robotics and Systems at HIT serves as the primary corresponding institution for this publication. Professor Zhang He from the School of Mechatronics Engineering (HIT), Assistant Professor Zhang Tianxue from Beihang University, and Doctor Jia Huan, director at the Ninth People's Hospital (Shanghai Jiao Tong University School of Medicine), are the co-corresponding authors. Doctor candidates Li Haiming and Gao Peiyuan from the School of Mechatronics Engineering (HIT), and Tan Haoyue, attending physician at the Ninth People's Hospital (Shanghai Jiao Tong University School of Medicine), are the co-first authors. Scholars from HIT, the Ninth People's Hospital (Shanghai Jiao Tong University School of Medicine), Beihang University, Sun Yat-sen University's Shenzhen campus, and Shandong University also contributed to this research.
This work was supported by the National Key R&D Program of China, the Distinguished Young Scholars Program of Heilongjiang Natural Science Foundation, the National Natural Science Foundation of China, and the Key Research and Development Program of Heilongjiang.
Paper link: https://www.nature.com/articles/s41467-026-72398-5