Harbin Institute of Technology Media (By Liu Peixiang, Zhou Wei, Yu Yang, Text/Images)
A research team led by Professor Zhou Wei from the group of Professor Gao Jihui at the School of Energy Science and Engineering, Harbin Institute of Technology(HIT), has made significant progress in understanding the dynamic protection mechanism at the catalytic interface for direct seawater electrolysis to produce hydrogen.
By in-situ monitoring the self-reconstruction of catalysts and the dynamic evolution of interfacial hydrogen bonding, the study reveals for the first time the correlation between catalyst reconstruction and stability, providing a novel design strategy for catalysts to overcome the challenge of chloride corrosion in seawater hydrogen production.
The research findings, titled Redefining catalyst reconstruction and Cl--repulsion correlation to delineate a dynamic protective skeleton for seawater splitting, have been published in Nature Communications.
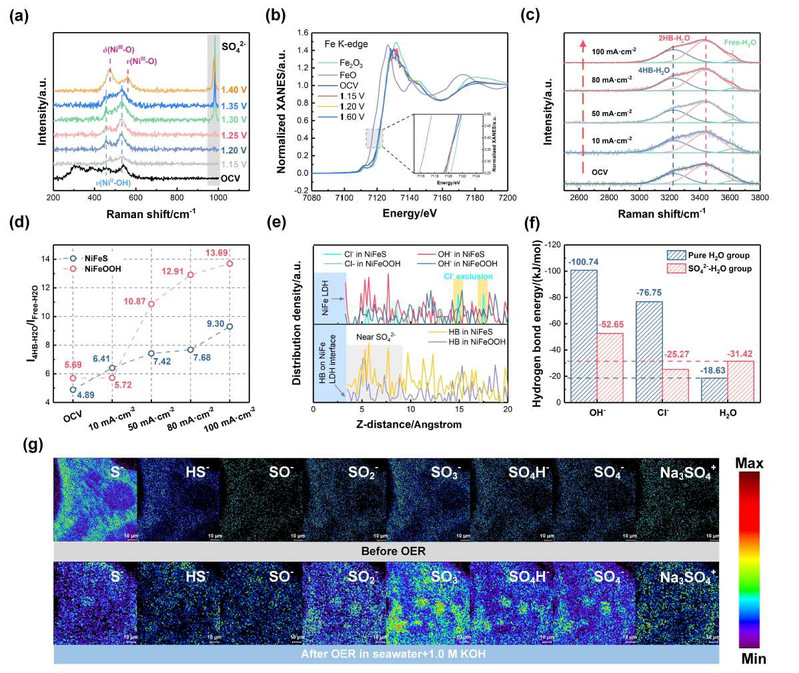
The mechanism of NiFeS catalyst reconstruction and the enhanced interfacial hydrogen-bond network for OH-/Cl- sieving.[Photo/hit.edu.cn]
Unlike laying high-cost submarine cables, seawater electrolysis for hydrogen production is considered a key technology for advancing offshore wind power. The corrosion issue induced by chloride ions (Cl-) in seawater is the primary obstacle hindering the practical application of direct seawater electrolysis. Previous studies have shown that in alkaline environments, the active reconstruction of catalysts not only generates active layered double hydroxides (LDH) but also forms a protective layer through the leaching of non-metallic elements, thereby enhancing catalyst stability. However, the underlying mechanism, particularly how anions participate in and reinforce interface stability, previously lacked direct evidence and a reasonable explanation at the atomic scale. This gap in theoretical understanding has limited the rational design of high-performance catalysts.
The research team employed a two-step Fe-S etching method to synthesize a large-area (1 m2) NiFeS catalyst. By combining in-situ Raman spectroscopy and in-situ synchrotron radiation spectroscopy, the team monitored the dynamic reconstruction process of NiFeS. Furthermore, AIMD simulations and in-situ Raman spectroscopy demonstrated that the SO42- formed after sulfur leaching effectively enhanced the interfacial hydrogen-bond network, promoting preferential OH- mass transport and inhibiting Cl- diffusion. The resulting highly active NiFe LDH and SO42- enabled highly stable electrolysis for 2000 hours at 1.0 A/cm2 in natural seawater and sustained operation over 1500 consecutive start-stop cycles. This study redefines the correlation between catalyst reconstruction and chloride corrosion resistance, emphasizing the critical role of catalyst reconstruction in stable seawater electrolysis, and provides an important foundation for designing catalysts and process routes for direct seawater electrolysis.
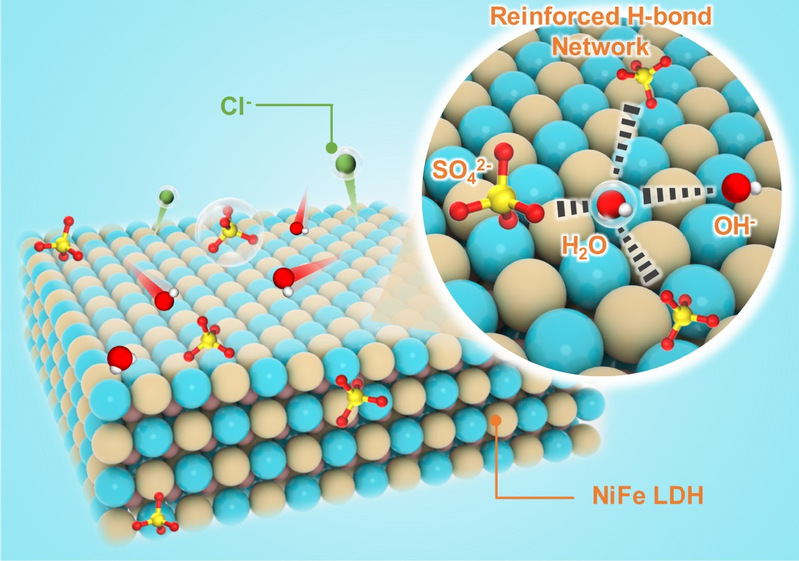
The schematic illustration of catalyst reconstruction and the OH⁻/Cl⁻ sieving mechanism.[Photo/hit.edu.cn]
In prior work, the team achieved stable seawater electrolysis for over a thousand hours at high current densities by regulating anode interfacial reactions and ion transport processes in alkaline seawater. The team is currently validating the stability of a 10-kilowatts prototype system based on this catalytic system. Relevant previous results have been published in journals such as Energy & Environmental Science, Advanced Functional Materials, ACS Catalysis, and ACS Sustainable Chemistry & Engineering, garnering widespread attention from peers both domestically and internationally.
The School of Energy Science and Engineering, HIT, is the sole corresponding unit for this paper. Doctoral candidate Yu Yang is the first author, and Professor Zhou Wei is the corresponding author. Doctoral candidates Zhou Xiaohan, Zhang Xuewei, Chen Yingjian, Master's candidate Yuan Junshu, Assistant researcher Li Xuhan, Associate professor Meng Xiaoxiao, Professor Sun Fei, Professor Gao Jihui, and Professor Zhao Guangbo, along with Postdoctoral researcher Xia Xiao from University of Jinan, Professor Zhang Liqiang from Shandong University, and Wang Xingxing from CHN Energy Yuedian Taishan Power Generation Co, participated in related research.
This work was supported by the National Energy Storage Technology Industry-Education Integration Innovation Platform, the National Natural Science Foundation of China, and the Heilongjiang Key R&D Program.
Paperlink : https://www.nature.com/articles/s41467-026-69755-9